Bioprocess Development & Protein Production
Navigating biologic production from discovery through bioprocess development to manufacturing presents a variety of challenges including low productivity, lack of scalability and extended timelines for stable cell line generation. High-efficiency transient transfection of virtually any cell type using the MaxCyte ExPERT™ platform enables the rapid production of an array of protein-based biologics including antibodies, recombinant proteins, vaccines, viral vectors and virus-like particles.
Flexibility to support more efficient bioprocess development
MaxCyte electroporation technology enables high-efficiency transfection for transient expression of multigram quantities of protein-based biologics and viral vectors.
- Delivery of diverse payloads including:
- Nucleic acids
- Proteins
- Small molecules
- High cell viability for faster production
- Seamless scalability with one transfection technology and your chosen manufacturing cell line
- Streamlined workflow with optimized reagents and protocols
- High-titer transient expression from gene to product in days or weeks, not months
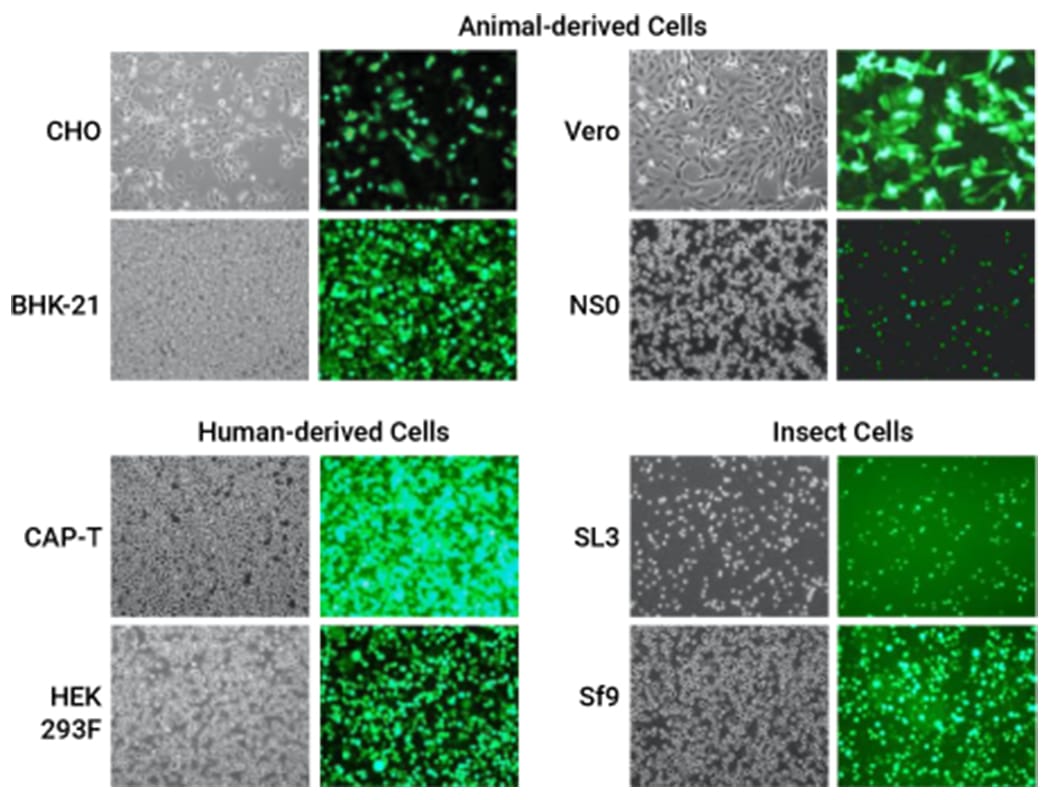
Efficient GFP expression following pGFP electroporation of commonly used cell types
Achieve high titers with your preferred manufacturing cell type to de-risk development
The MaxCyte ExPERTTM platform gives you the freedom to use the cell type that makes the most sense for your project.
MaxCyte’s electroporation technology delivers high transfection efficiency and cell viability for high titer transient production in your manufacturing cells. This means you don’t have to risk derailing your project when switching from an R&D cell line.
MaxCyte’s ExPERT platform enables reproducible transfection of many cell types commonly used for protein production including:
- CHO and CHO-derivatives
- HEK293
- Vero
- NS0
- Insect
- SP2/0
Enable lead progression in parallel with stable cell line development
A single, large scale electroporation with MaxCyte’s STx or VLx transfects a sufficient number of cells to split between batch culture and stable cell line development. Generating grams of purified protein or complex biologics by transient expression enables you to start chemistry, manufacturing and controls, as well as toxicology studies, within weeks of candidate identification instead of waiting months for material from a stable cell line. Because when speed to the clinic matters, every day of development time you save counts.
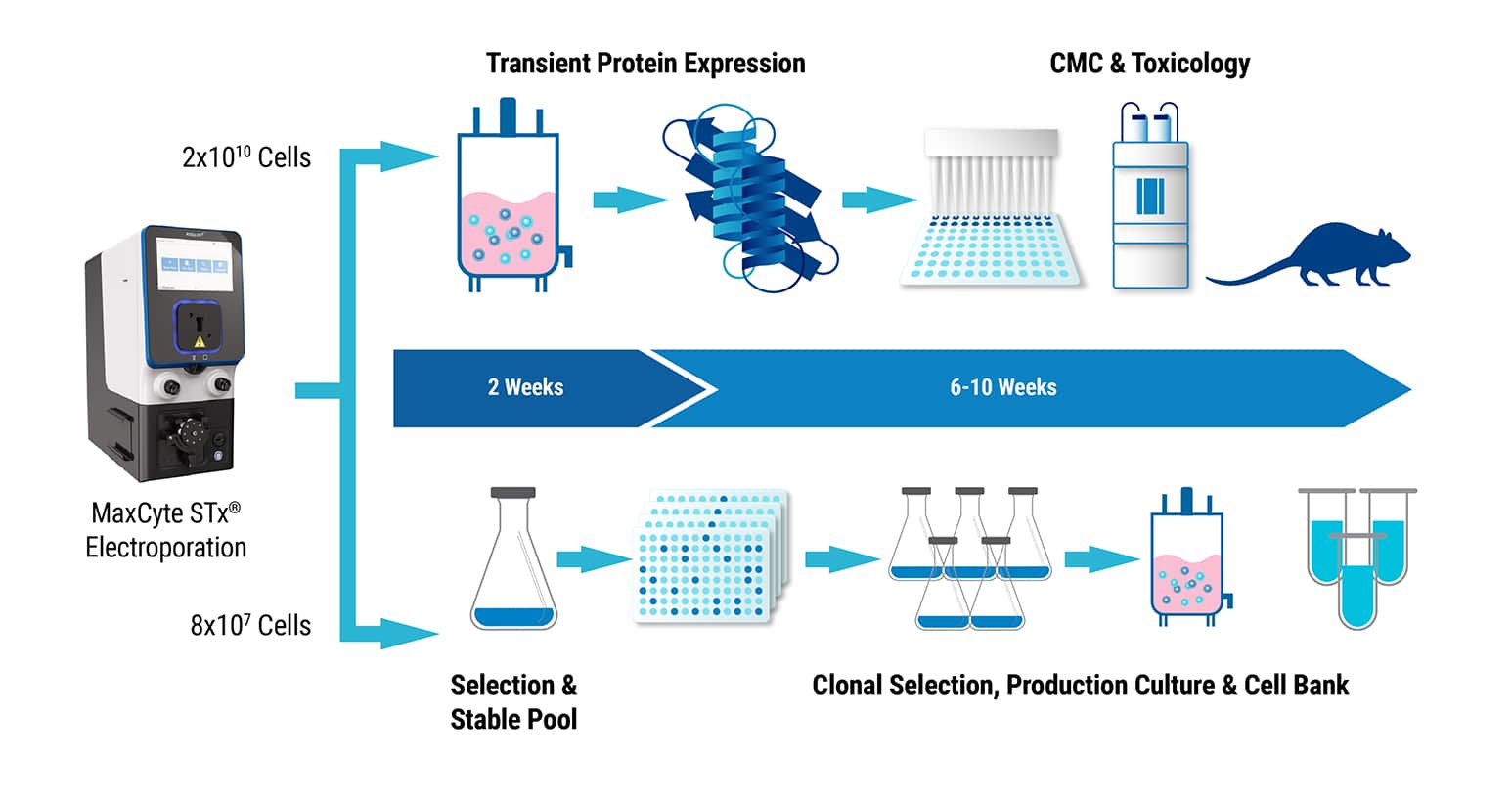
Research applications
Resources
Ready to learn more about enabling bioprocessing and protein production?
Answers are just a click away!







