Our Company
MaxCyte® is a leading provider of cell-engineering technologies advancing innovative cell-based research, clinical development, and commercialization of next‐generation cell therapies. We are focused on making a meaningful difference in the lives of patients and have spent over 20 years optimizing, innovating, and collaborating to enable scientists and doctors with the best-in-class electroporation technology.
MaxCyte’s electroporation technology is used by leading drug developers, hundreds of biotechnology companies and academic translational centers worldwide. We have also established partnerships with 23 leading biotech companies. (link to partner page) Our technology has enabled the manufacturing of drug products for over 45 clinical trials and leveraged in over 125 clinical programs, expanding over a range of diseases and illnesses. To date, there are over 1,000 patients enrolled and participating in active clinical trials empowered by MaxCyte technology.
MaxCyte® securities are traded on the AIM Market, operated by the London Stock Exchange and the Nasdaq Global Select Market, with ownership largely controlled by institutional shareholders.

Our Approach
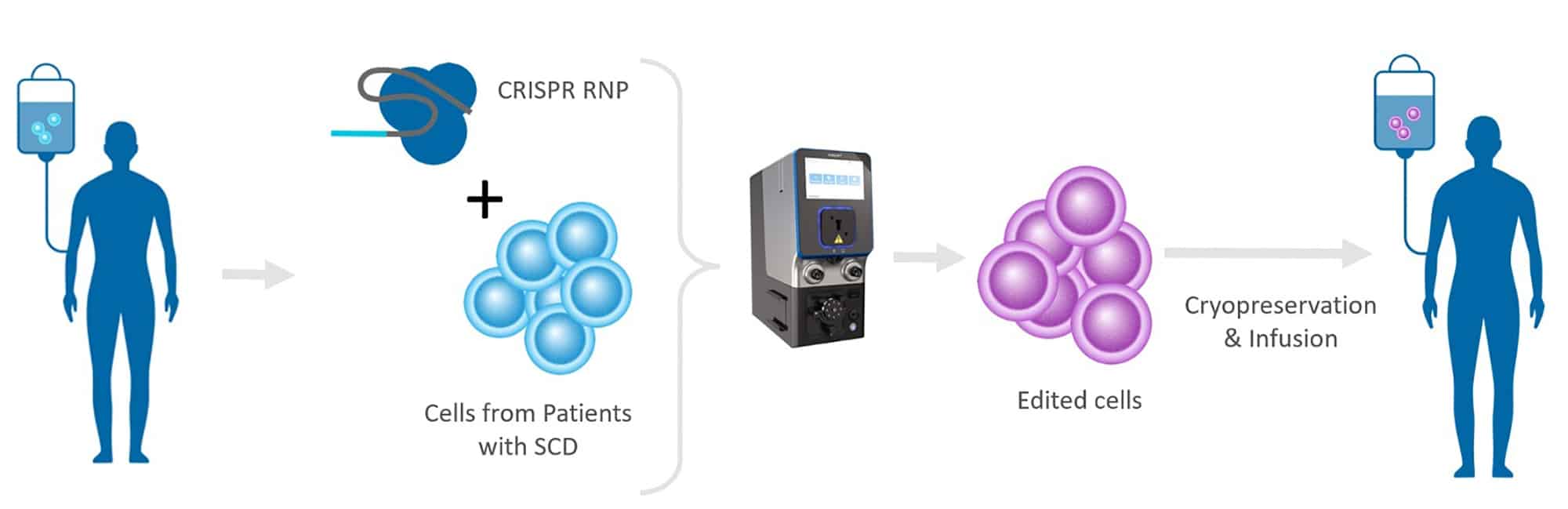
Electroporation is a method that applies an electric pulse to cells, which causes cell membranes to become temporarily permeable. Diverse genetic engineering tools such as DNA, RNA, CRISPR, and Ribonucleoproteins (RNPs) can then travel through the membrane and enter the cell. Electroporation is a highly efficient delivery method that is used in in a wide range of applications including scientific research, clinical trials and now in a marketed product for the treatment of sickle cell disease (SCD).
Value for Patients Initiative
In addition to providing the sector with a best-in-class technology we are also committed to understanding how we, at MaxCyte can create value for patient communities. We are keenly aware that many barriers exist in healthcare access, equity, and adoption. In particular, we are dedicated to understanding the unique challenges associated with cell and gene therapy.
We launched our Value for Patients Initiative in 2021 to better understand the difficulties that discrete patient populations may face with current standards of care and forthcoming cell- and gene-based medicines. As part of this initiative, we have developed relationships with key opinion leaders who are helping us to understand the social, racial, economic, and bioethical challenges related to developing cell and gene therapies. In this way, we proactively participate in efforts to ensure that these novel treatments reach patients.
In 2023, with the support of our advisors, we developed an internal educational program to empower our employees, leadership and board of directors to better understand various patient communities. This educational series covered topics including medical racism, equity of access to medicines, clinical trial diversity, compassionate use of cell and gene therapy, and other issues affecting patient experiences.

Our Advisors

Amanda Calhoun, MD
Adult/Child Psychiatry
Resident at Yale University
School of Medicine

Arthur L. Caplan, PhD
Mitty Professor and
founder Division of Medical
Ethics at NYU School of
Medicine

Vivien Sheehan, MD, PhD
Director of Translational
Research and Associate
Professor of Pediatrics at
Emory University
Interactive Workshops 2023

2023 Session 1
"The Sickle Cell Patient Journey"
The overall goal of this first session in the Value for Patients Series was to provide an interactive forum with Dr. Sheehan to learn about life journey of a SCD patient, the disease stages, the treatment options, the challenges, and the choices these patients face.

2023 Session 2
“Why does Cell and Gene Therapy Command so much Ethical Attention?”
In the 2nd session, we became keenly aware that many challenges exist in healthcare access, equity, and adoption, including the unique barriers patients face in accessing the benefits of cell and gene therapies.

2023 Session 3
“Patient Trust is Earned”
Building and maintaining trust with patient communities of color requires that we:
Shaping Genetic Medicines
Since the 1960s and the discovery of DNA, scientists have envisioned using this genetic material to treat genetic disorders. The concept of gene therapy was introduced in the late 1970s following the advancement of recombinant DNA technology. Subsequent to the establishment of fundamental scientific principles and technologies for transferring genes into patients' cells, the initial human gene therapy trial took place in 1990. By 2003, the completion of the Human Genome Project provided a full blueprint of our DNA offering essential insights into the human blueprint. This information has propelled the exploration of human biology and enhanced the field of medicine over the past decade, making gene therapy a tangible solution for various diseases, particularly those resulting from mutations in a single gene.
What is Cell and Gene Therapy?
Cell and gene therapies constitute a distinctive category of medical technologies designed to address diseases by introducing either entire cells (cell therapy) or genetic material (gene therapy) into patients. While these approaches share some common ground, there are notable distinctions between cell and gene therapies.
Gene therapy involves the incorporation of new genetic material, or the modification of defective genes linked to a specific disease in a patient. These modifications can occur either directly within the patient's body (in vivo) or through the manipulation of cells outside the body (ex vivo). With over 5,000 recognized monogenic diseases—conditions caused by mutations in a single gene—gene therapy holds promise in correcting these genetic anomalies or supplying a functional gene copy to improve the associated conditions.
On the other hand, cell therapy entails infusing a patient with healthy cells to address deficiencies or replace diseased cells. Cell therapy can be autologous, where the cells are sourced from the patient, or allogeneic, involving cells derived from healthy donors.
1953
DNA Discovery
The structure of DNA is characterized by a double helix.2.
1973
Engineering DNA
Researchers discover genetic engineering.
1990
First clinical trial using viral vector technology
Two patients with a rare inherited pediatric disorder called severe combined immunodeficiency (SCID) receive treatment using novel gamma-retroviral vector technology.
1996
The first engineered nuclease technology (zinc finger nuclease) for exploring the use of zinc finger nucleases for gene editing as a potential for gene therapy.
1999
Setbacks in research
The US Food and Drug Administration (FDA) and NIH create new programs in an eff¬ort to ensure the safety and transparency of gene therapy clinical trials following the death of Jesse Gelsinger, an 18-year-old patient, during a clinical trial using an adenoviral vector.
2003
Human Genome Project completed
2010
The first engineered TAL-effector nucleases (TALEN) were described with the ability to cause targeted mutagenesis.
2012
The European Medicines Agency (EMA) approved the first AAV-based gene addition therapy for the treatment of lipoprotein lipase deficiency (LPLD) in vivo – Glybera™.
2012
At the University of Pennsylvania, Emily Whitehead was the first pediatric patient in the world to receive CAR T-cell therapy.
2012
Discovery of CRISPR Cas9
2014
First clinical application of genome editing involved the use of ZFNs to make human cells resistant to HIV-1 by disrupting a gene required for the virus to infect cells.
2016
The EMA approved the first gamma retrovirus-based gene addition therapy to treat adenosine deaminase severe combined immunodeficiency (ADA-SCID). This therapy contains CD34+ cells transduced with retroviral vector, which encodes for the human ADA cDNA sequence – ex vivo viral-based – Strimvelis™.
2017
First gene therapy approval in the US The FDA approves the first AAV-based in vivo gene addition therapy for a genetic eye disease that causes progressive vision loss. – Luxturna ™.
2017
FDA approval of first viral-based CAR T cell therapies Yescarta™ and Kymriah™.
2018
First clinical trial using CRISPR/Cas9 Researchers investigate the use of CRISPR/Cas9 for gene disruption in patients with genetic β-hemoglobinopathies.
2019
The FDA approved an AAV-based in vivo gene addition therapy for spinal muscular atrophy – Zolgensma™.
2019
The EMA approved an LVV-based ex vivo gene addition therapy for a genetic disease called transfusion-dependent beta-thalassemia (TDT) Zynteglo™.
2020
Victoria Gray became the first patient to be treated for sickle cell disease using CRISPR/Cas9, an experimental gene-editing technique.
2022
U.S. Food and Drug Administration approved Zynteglo™ (betibeglogene autotemcel), the first cell-based gene therapy for the treatment of adult and pediatric patients with beta-thalassemia.
2023
United Kingdom (U.K.) Medicines and Healthcare products Regulatory Agency (MHRA) has granted conditional marketing authorization for CASGEVY™ (exagamglogene autotemcel [exa-cel]), a CRISPR/Cas9 gene-edited therapy, for the treatment of sickle cell disease (SCD) and transfusion-dependent beta thalassemia (TDT).
2023
CASGEVY™ Gets Bahrain approval for treatment, marking it second country in the world https://www.globenewswire.com/news-release/2023/12/02/2789644/0/en/CASGEVY-Gets-Bahrain-approval-for-treatment-marking-it-second-country-in-the-world.html.
2023
CASGEVY™ FDA Approval
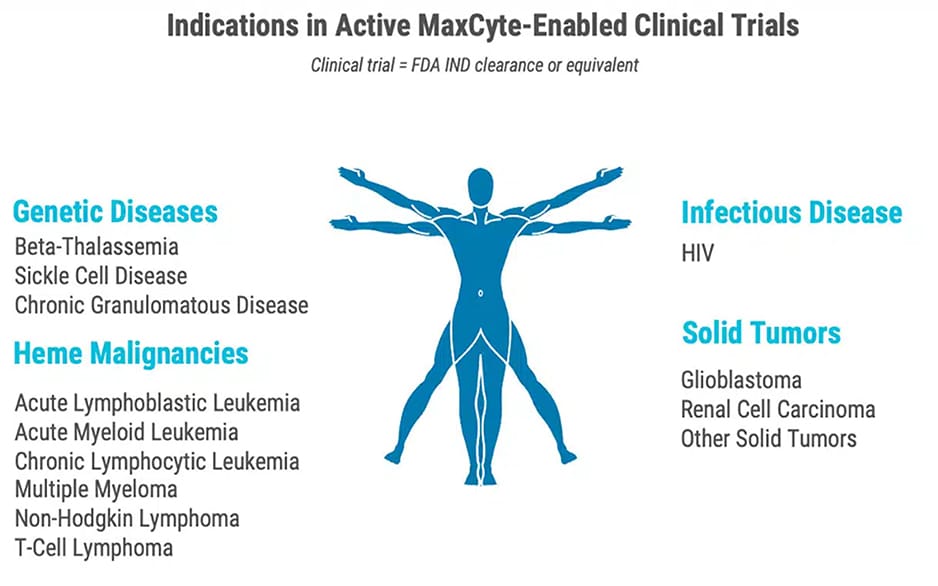
Supporting Innovation and Therapy Development for A Variety of Indications
At MaxCyte, we recognize that it takes much more than innovative technology to bring a new therapy to market. While MaxCyte is not a therapeutic developer, we create gene-engineering tools that enable our biopharma partners to develop transformative therapies.
Our goal is to rapidly drive our partners’ development efforts forward through to commercialization cost-effectively and with lowered risk.
We strive to expand access to cell therapies for all patients, regardless of disease or disorder. A recent study estimated that there are over 10,000 distinct rare diseases affecting 400 million people worldwide, yet only 500 of these disorders have available treatment options. We support organizations developing treatments for large indications and companies developing therapies to treat rare and ultra-rare diseases with few or no treatments.
Engineered cell therapies for Sickle Cell Disease
Millions worldwide carry the gene responsible for sickle cell disease (SCD). Although millions of people around the world live with a type of sickle cell disease, it is still considered a rare condition in the U.S., affecting an estimated 100,000 people. In people with sickle cell, a genetic mutation causes the cells to become crescent-shaped, which can block blood flow and cause excruciating pain, organ damage, stroke and other problems. The fact that a single mutation usually causes SCD makes it an attractive target for genetic editing.
Using MaxCyte’s electroporation technology, our therapeutic partners have been investigating various gene editing approaches to discover and develop potential treatments for SCD. These transformative therapies will offer hope to thousands affected by this crippling disorder.