Our Technology
Navigating the best route to development of groundbreaking cell therapies is an exciting challenge. At MaxCyte®, we’ve spent over 20 years honing our cell engineering expertise, optimizing static electroporation and innovating to create our Flow Electroporation® technology. We’ve perfected the art of the transfection workflow. Our collection of ExPERT™ instruments delivers engineering excellence, combining high efficiency and cell viability with seamless scalability. Our technology is empowering research journeys from concept to commercialization.

Advances in electroporation technology
We have spent over 20 years optimizing and innovating electroporation to bring you best-in-class electroporation technology, including:
- Electroporation buffer for all cell types
- Over 100 protocols for a variety of cell types
- Improved cuvette design with our Processing Assemblies
- Groundbreaking Flow Electroporation protocol to enable cGMP-compliant, large-scale genetic engineering
- Connectivity for closed system cGMP manufacturing
Understanding Flow Electroporation
Electroporation is a non-viral transfection technique that uses electricity to relax cell membranes, allowing payload to enter. Our Flow Electroporation technology is uniquely designed to allow cells to flow through the processing chamber where discrete volumes are electroporated, then collected on a continual basis. This pioneering innovation makes genetic engineering at large scale a reality.

Load any molecule at any scale in any cell
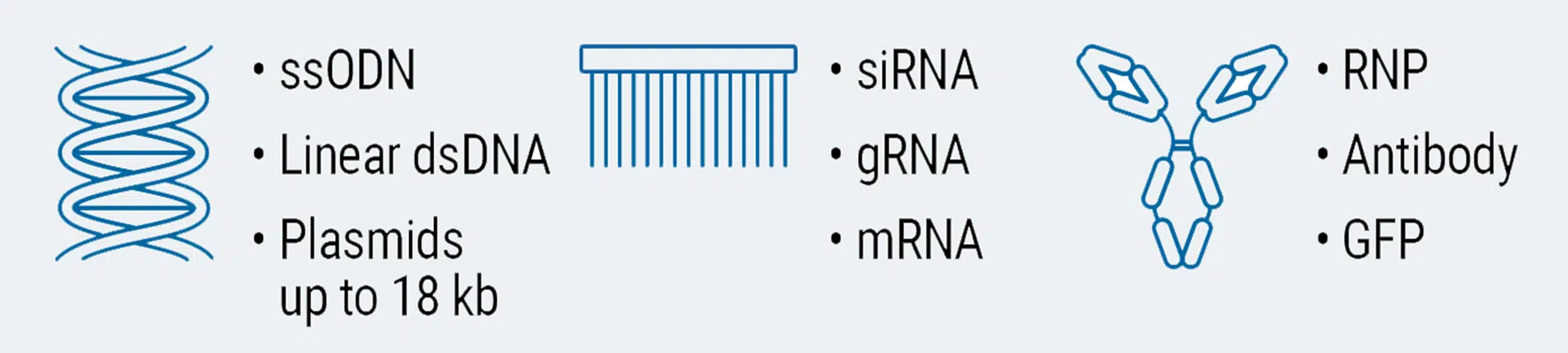
Cells and Scalability
Whether you are at the beginning of the research journey transfecting thousands of cells or in the middle of scale-up looking to transfect billions of cells, MaxCyte is the best partner to help steer your project to success with our scalable electroporation technology.
With over 100 proprietary, optimized electroporation protocols for a broad range of cell types we’ve got you covered wherever your journey of discovery takes you!
Streamlined therapeutic development
MaxCyte has evolved the electroporation process from a single cuvette experiment to a Flow Electroporation® protocol ideal for genetic engineering at commercial scale. Higher transfection efficiency and increased cell viability compared to other approaches make Flow Electroporation perfectly suited for large-scale therapeutic manufacturing. And because the complete workflow, from development to manufacturing, can be performed on a single platform, there is no need for repeat optimization and validation when progressing from concept to clinic. This results in accelerated timelines with the added benefit of reduced manufacturing cost. A more rapid, reliable and safe therapeutic development pipeline can expedite your therapies to patients, saving lives.
Resources
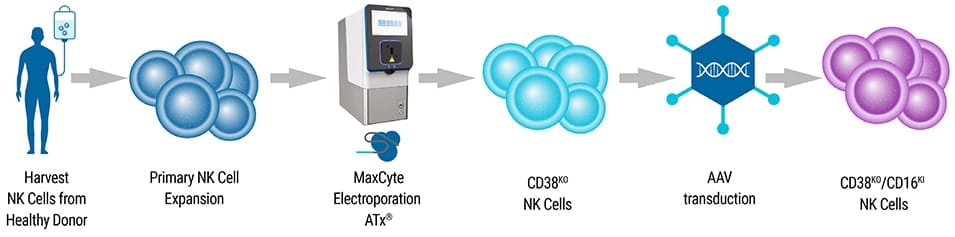
Application Note
MaxCyte® Enables Multiplexed NK Cell Engineering.
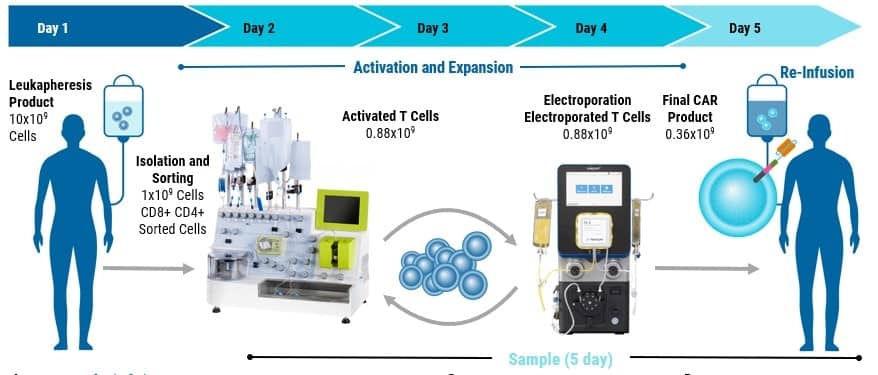
Poster
Electroporation of a Non-Integrative DNA Nanovector for Efficient, Semi-Automated, GMP Manufacturing of CAR T Cell Therapies.
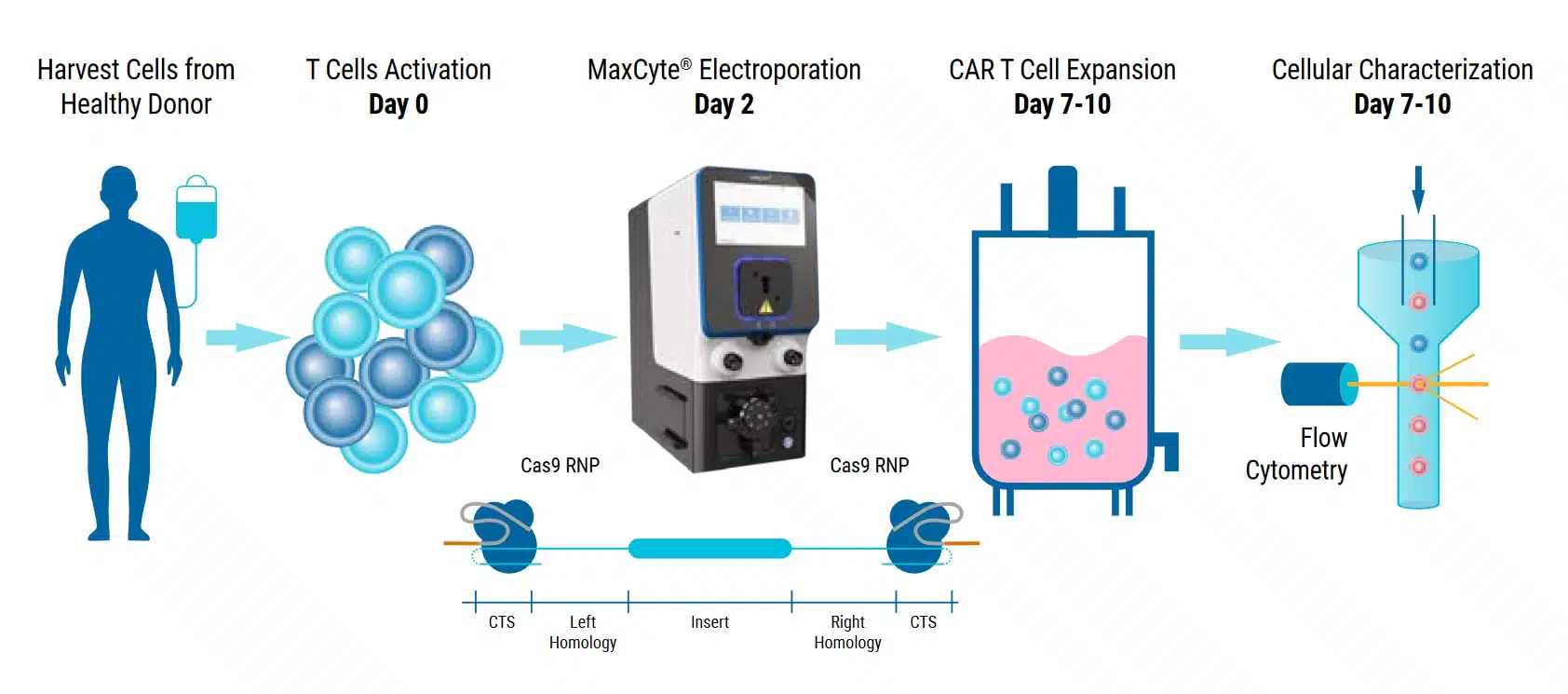
Application Note
MaxCyte® Enables a cGMP-Compatible Manufacturing Process for Highly Efficient T Cell Engineering.
Electroporation Systems
Supported by numerous publications and clinical trials, ExPERT instruments are the next generation of industry-leading scalable electroporation technology.
Our instruments are capable of high-performance delivery of virtually any molecule, into any cell, at any scale. They transfect primary cells, stem cells and cell lines, all with high cell viability and transfection efficiencies routinely over 90%.


Reagents and Processing Assemblies
MaxCyte’s consumable products provide a variety of options for project scale and throughput from discovery through cGMP manufacturing using a single platform. Our range of Processing Assemblies allows users to transfect a variety of cell sample volumes to meet specific application needs. MaxCyte’s Electroporation Buffer is animal-derived component free and safe for all cell types ensuring consistent, high-performance transfection.
Our customers need more than just technology to achieve their goals.
They need the unique combination of tools and resources we provide.
The three pillars at the very core of our company are:
Ready to learn more about our technology?
Find out how our personalized support can ease your journey to the clinic.





