KSQ Achieves IND Clearance for Solid Tumor Treatment with MaxCyte Support
In this case study, KSQ Therapeutics, a clinical-stage company developing engineered T cell immunotherapies for solid tumors, leverages MaxCyte’s advanced cell engineering platforms to ultimately gain clinical clearance.
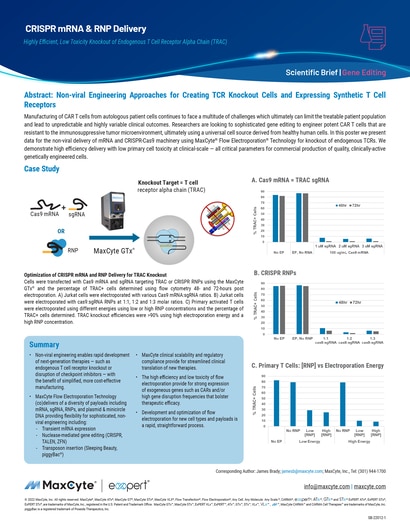
Read MoreCase Study: CRISPR mRNA & RNP Delivery
Demonstrating high efficiency delivery with low primary cell toxicity at clinical-scale — all critical parameters for commercial production of quality, clinically-active genetically engineered cells.
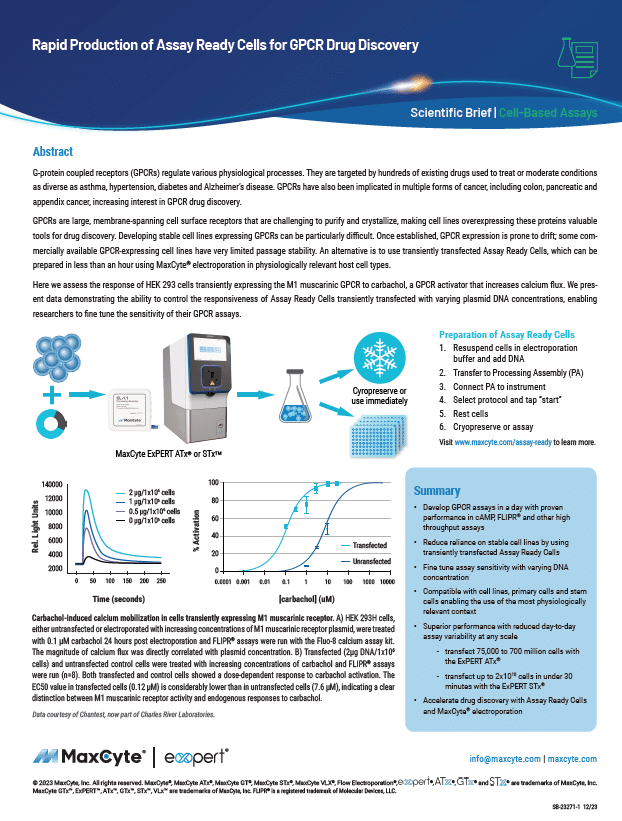
Read MoreRapid Production of Assay Ready Cells for GPCR Drug Discovery
G-protein coupled receptors (GPCRs) regulate various physiological processes. They are targeted by hundreds of existing drugs used to treat or moderate conditions as diverse as asthma, hypertension, diabetes and Alzheimer’s disease. GPCRs have also been implicated in multiple forms of cancer, including colon, pancreatic and appendix cancer, increasing interest in GPCR drug discovery.
Read MoreEx Vivo Gene Correction in X-CGD Patient Stem Cells
Discover how MaxCyte® Flow Electroporation® enabled efficient gene correction in patient-derived CD34+ hematopoietic stem cells through the delivery of CRISPR-Cas9. Explore how engraftment of edited cells in mouse peripheral blood and bone marrow was achieved.
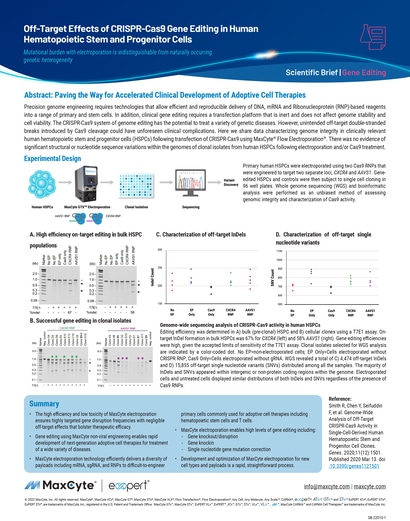
Read MoreOff-Target Effects of CRISPR-Cas9 Gene Editing in Human Hematopoietic Stem and Progenitor Cells
Here we share data characterizing genome integrity in clinically relevant human hematopoietic stem and progenitor cells (HSPCs) following transfection of CRISPR-Cas9 using MaxCyte® Flow Electroporation®.
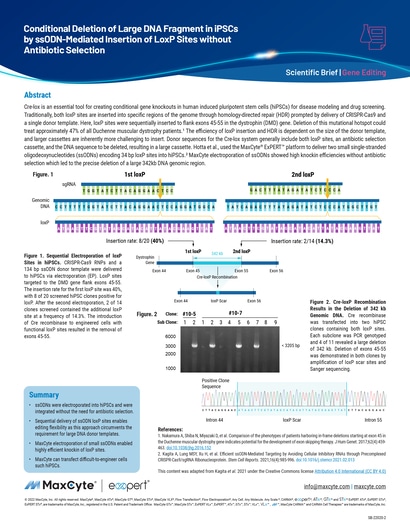
Read MoreConditional Deletion of Large DNA Fragment in iPSCs by ssODN-Mediated Insertion of LoxP Sites without Antibiotic Selection
Cre-lox is an essential tool for creating conditional gene knockouts in human induced pluripotent stem cells (hiPSCs) for disease modeling and drug screening. Traditionally, both loxP sites are inserted into specific regions of the genome through homology-directed repair (HDR) prompted by delivery of CRISPR-Cas9 and a single donor template.
Read MoreIgnite Your Cell Therapy Development with MaxCyte ExPERT Electroporation Platform
Whether you’re developing allogeneic or autologous cell therapies, getting efficient transfection can save time and precious patient samples, moving you into the clinic faster. With the ExPERT platform you can rely on high transfection efficiency with any cell type or transfected molecule.
Read MoreThe ExPERT ATx is capable of high-performance delivery of virtually any molecule, into any cell
Hold your research and proof-of-concept studies to the highest standards with reliable and efficient electroporation technology.
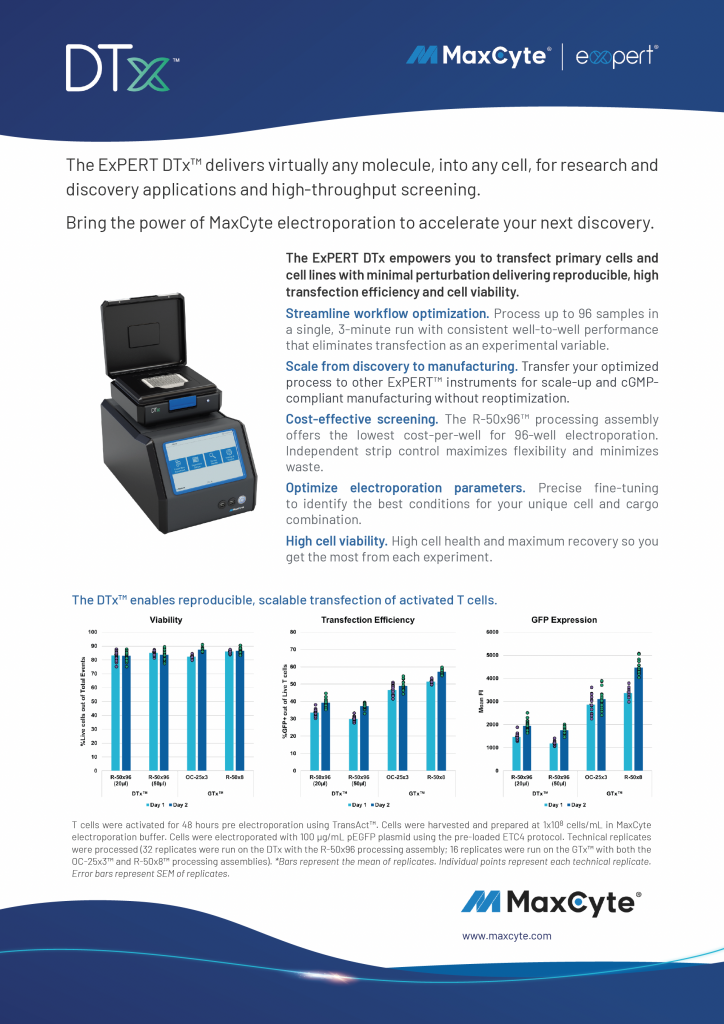
Read MoreThe ExPERT DTx
Streamline 96-well electroporation with consistent performance and precise parameter optimization while maintaining high cell viability. Seamlessly scale optimized workflows across ExPERT instruments from discovery to cGMP manufacturing in a flexible, cost-effective format.
Read MoreThe ExPERT GTx
The ExPERT GTx provides enhancements that improve ease of use, processing workflow, regulatory compliance and overall user experience with its elegant design that fits seamlessly into any high-tech laboratory space
Read More