MaxCyte® Flow Electroporation Technology: A Safe, Reliable and Effective Method for Engineering CAR T Cells
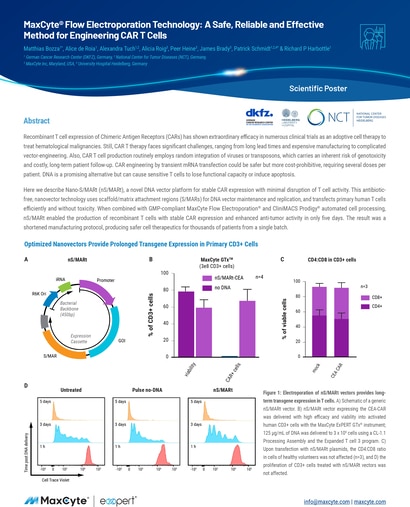
Recombinant T cell expression of Chimeric Antigen Receptors (CARs) has shown extraordinary efficacy in numerous clinical trials as an adoptive cell therapy to treat hematological malignancies.
Read MoreMaxCyte® Enables Multiplexed NK Cell Engineering
As the immune system’s first responders, natural killer (NK) cells are essential for the rapid detection and elimination of cancer. Their ability to kill cancer cells without prior sensitization makes them an ideal immunotherapy candidate.
Read MoreAchieving Therapeutic Efficacy with a High Performance, Electroporation-based Cellular Engineering Technology
Advances in cell-based therapies for treating cancer and other diseases have created a demand for rapid and flexible manufacturing processes.
Read MoreHighly Efficient Homozygous Correction of the DYSF Gene in Miyoshi Myopathy Patient iPSCs by ssODN-Mediated Knockin
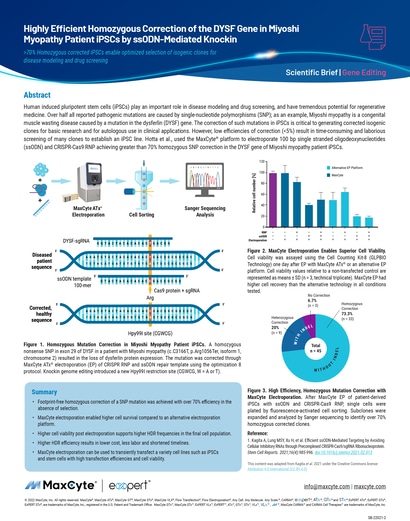
Human induced pluripotent stem cells (iPSCs) play an important role in disease modeling and drug screening, and have tremendous potential for regenerative medicine.
Read MoreGeneration of Heterozygous HLA-C iPSCs by ssODN Electroporation for Allogeneic Transplantation
Human-induced pluripotent stem cells (iPSCs) have tremendous potential for regenerative medicine. While there is an ongoing effort to recruit human leukocyte antigen (HLA) homozygous donors for allogeneic transplantation, a significant challenge with this approach is the susceptibility of these cells to host natural killer (NK) cell cytotoxicity.
Read MoreAdvancing a CCR5 Gene Editing Therapy to Clinical Trial: Overcoming Viral Vector Toxicity
HIV infection is currently managed by lifelong antiretroviral therapy (ART), a modality that is associated with chronic toxicity, challenging patient compliance, and comes at a significant cost over a patient’s lifetime.
Read MoreBoosting the Efficacy of Tumor Infiltrating Lymphocytes via PD-1 Gene Editing
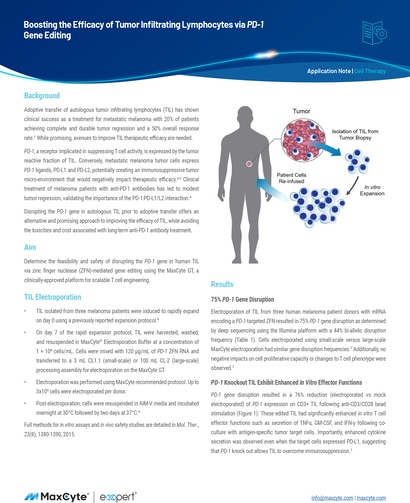
Adoptive transfer of autologous tumor infiltrating lymphocytes (TIL) has shown clinical success as a treatment for metastatic melanoma with 20% of patients achieving complete and durable tumor regression and a 50% overall response rate.1 While promising, avenues to improve TIL therapeutic efficacy are needed.
Read MoreAdvancing an HIV Clinical Program for CCR5 Gene Disruption
Precision genome engineering requires technologies that allow efficient and reproducible delivery of DNA, mRNA and RNP-based reagents into a range of primary cells and stem cells. In addition, clinical gene editing requires a transfection platform that is GMP-compliant and scalable to accom- modate billions of cells in a single transfection.
Read MoreGene Knockin via Homology-directed Repair in Human iPSCs
Clinically validated and scalable MaxCyte® Flow Electroporation® Technology delivered ribonucleoprotein (RNP) and single-stranded oligonucleotide donor (ssODN) to induced pluripotent stem cells (iPSCs).
Read MoreCase Study: Investing In Your Success – Kamau Therapeutics Supported by MaxCyte
Kamau Therapeutics is a clinical-stage gene correction company advancing cell therapy development with high-efficiency targeted gene integration. It partnered with MaxCyte to optimize cell engineering processes and advanced its lead program, nula-cel, currently in clinical development for Sickle Cell Disease.
Read More