The ExPERT STx Instrument Delivers Virtually Any Molecule at Any Scale into Any Cell
The ExPERT STx is the industry’s leading scalable electroporation technology for high-yield transient expression of complex proteins, vaccines and biologics.
Read MoreIntroducing the VLx System
Achieve superior transfection efficiency and cell viability at scale with the ExPERT VLx and MaxCyte’s Flow Electroporation technology.
Read MoreAccelerate Antibody Development and Production
The MaxCyte ExPERT electroporation platform, supported by a global team of R&D and application scientists, can help accelerate your therapeutic antibody development and discovery to bring new treatments to patients sooner.
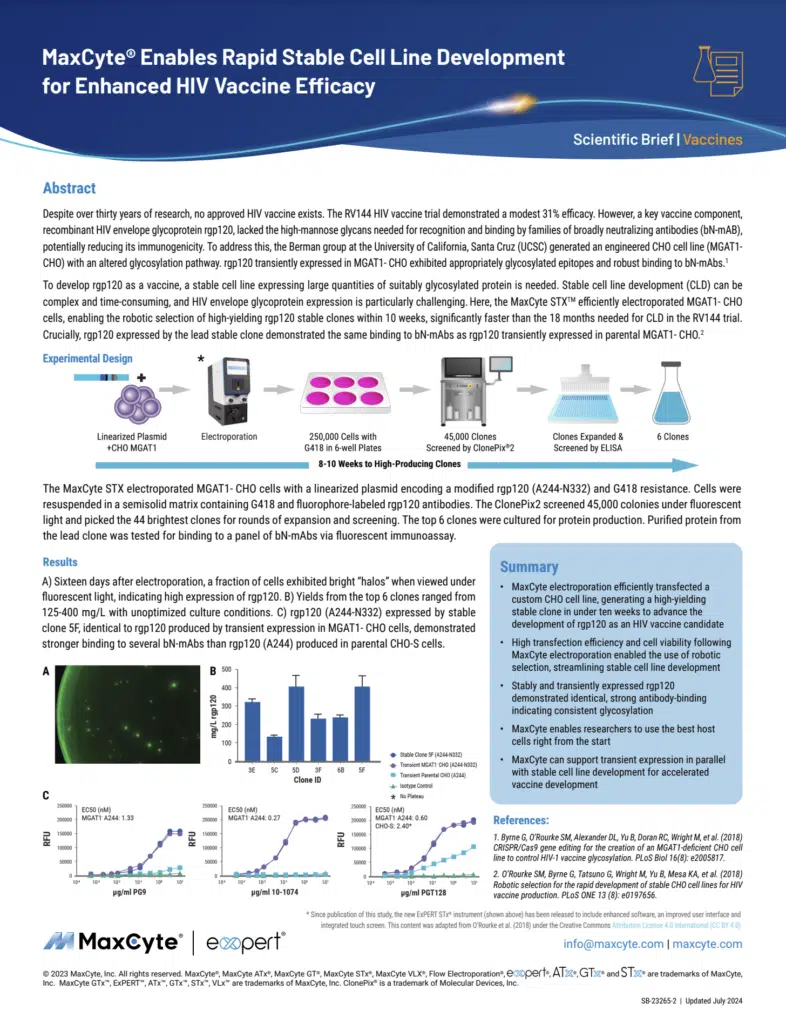
Read MoreMaxCyte Enables Rapid Stable Cell Line Development for Enhanced HIV Vaccine Efficacy
Despite over thirty years of research, no approved HIV vaccine exists. The RV144 HIV vaccine trial demonstrated a modest 31% efficacy. MaxCyte electroporation efficiently transfected a custom CHO cell line, generating a high-yielding stable clone in under ten weeks to advance the development of rgp120 as an HIV vaccine candidate.
Read MoreMaxCyte Electroporation Enables the Efficient, Scalable Packaging of CRISPR-Cas9 RNPs in Virus-Like Particles
Efficient, scalable cGMP-compliant transfection of up to 100 billion cells with MaxCyte’s Flow Electroporation technology could enable rapid clinical manufacturing of virus-like particles.
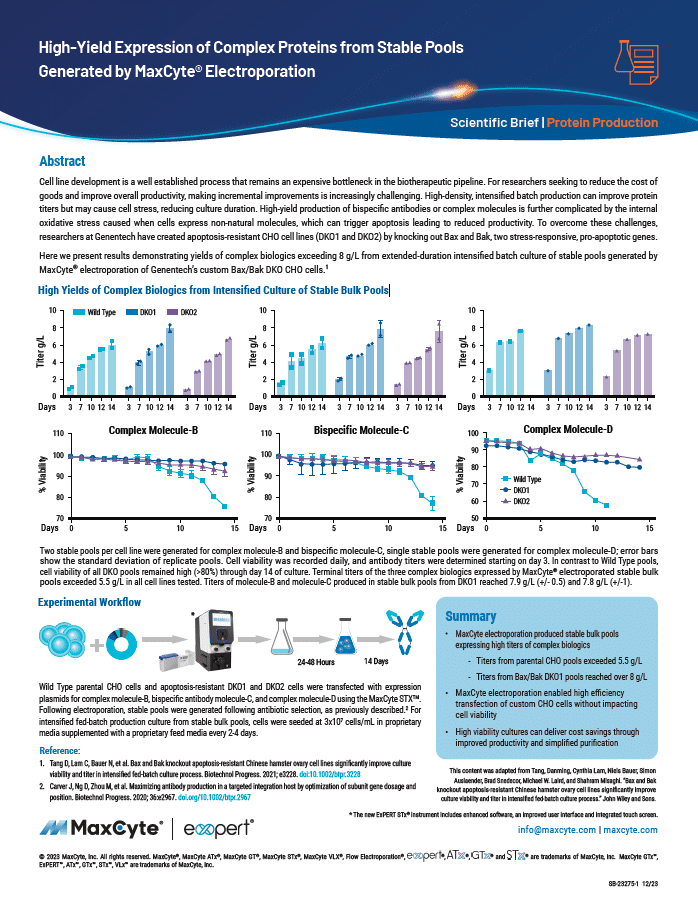
Read MoreHigh-Yield Expression of Complex Proteins from Stable Pools Generated by MaxCyte Electroporation
Cell line development is a well established process that remains an expensive bottleneck in the biotherapeutic pipeline. Here we present results demonstrating yields of complex biologics exceeding 8 g/L from extended-duration intensified batch culture of stable pools generated by MaxCyte electroporation of Genentech’s custom Bax/Bak DKO CHO cells.
Read MoreEnhancing HIV-1 Vaccine Efficacy and Manufacturing Through CHO Cell CRISPR Engineering
The RV144 HIV vaccine trial, the first trial to demonstrate that vaccination could confer HIV protection, reported a modest 31% efficacy. gp120, a major component of the RV144 trial, was produced in Chinese Hamster Ovary (CHO) cells and lacked N-linked glycosylation sites critical for binding of anti-HIV broadly neutralizing antibodies (bN-mAbs).
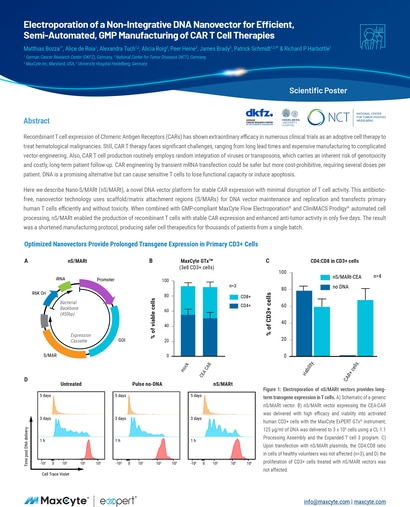
Read MoreElectroporation of a Non-Integrative DNA Nanovector for Efficient, Semi-Automated, GMP Manufacturing of CAR T Cell Therapies
Recombinant T cell expression of Chimeric Antigen Receptors (CARs) has shown extraordinary efficacy in numerous clinical trials as an adoptive cell therapy to treat hematological malignancies.
Read MoreEfficient and Scalable T Cell Engineering for Advancing Cancer Immunotherapy of Hepatocellular Carcinoma by a cGMP-Compliant Non-Viral Cell Engineering Platform
Chronic hepatitis B virus (HBV) infection can result in hepatocellular carcinoma (HCC), which is the third leading cause of cancer-related deaths worldwide.
Read MoreEngineering NK Cells with High-Affinity CD16 for Enhanced Combination Therapy to Treat B-Cell Lymphoma
NK cells play a pivotal role in rapid and highly efficient cancer surveillance and represent a promising avenue for adoptive cell transfer either as a mono or combination immunotherapy.
Read More