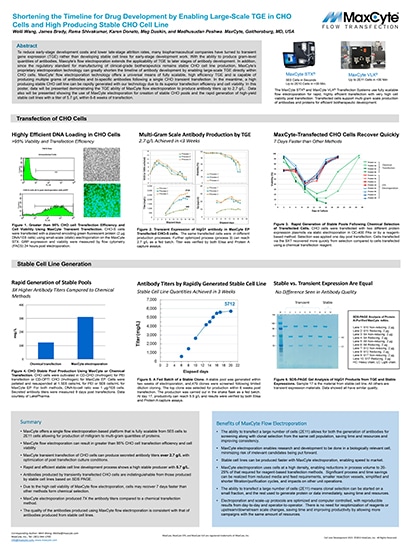
Shortening the Timeline for Drug Development by Enabling Large-Scale TGE in CHO Cells and High Producing Stable CHO Cell Line
To reduce early-stage development costs and lower late-stage attrition rates, many biopharmaceutical companies have turned to transient gene expression (TGE) rather than developing stable cell lines for early-stage development work.
Read MoreSimple and Effective Generation of Cell-based Assays for Ion Channels, Transporters, and Kinase Screening in Biologically Relevant Cells Using Scalable Transient Transfection
Cell-based assays utilizing mammalian cells to express receptor targets are widely used to rapidly identify molecular mechanisms of human disease and to develop novel therapeutics.
Read MoreSingle-Dose Intranasal Administration of AdCOVID Elicits Systemic and Mucosal Immunity against SARS-CoV-2 and Fully Protects Mice from Lethal Challenge
Intranasal vaccination is an attractive strategy to prevent COVID-19 as the nasal mucosa represents the first-line barrier to SARS-CoV-2 entry.
Read MoreStructure of human DPEP3 in complex with the SC-003 antibody Fab fragment reveals basis for lack of dipeptidase activity
Journal of Structural Biology – Dipeptidase 3 (DPEP3) is one of three glycosylphosphatidylinositol-anchored metallopeptidases potentially involved in the hydrolytic metabolism of dipeptides. While its exact biological function is not clear, DPEP3 expression is normally limited to testis, but can be elevated in ovarian cancer.
Read MoreTargeted gene addition in human CD34+ hematopoietic cells for correction of X-linked chronic granulomatous disease
Gene therapy with genetically modified human CD34+ hematopoietic stem and progenitor cells (HSPCs) may be safer using targeted integration (TI) of transgenes into a genomic ‘safe harbor’ site rather than random viral integration.
Read MoreTreatment of Advanced Leukemia in Mice with mRNA Engineered T Cells
Cytotoxic T lymphocytes (CTLs) modified with chimeric antigen receptors (CARs) for adoptive immunotherapy of hematologic malignancies are effective in preclinical models and are being tested in several clinical trials. Although CTLs bearing stably expressed CARs generated by integrating viral vectors are efficacious and have potential long-term persistence, this mechanism of CAR expression can potentially result in significant toxicity.
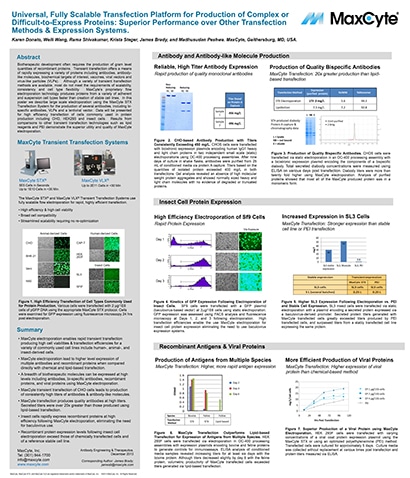
Read MoreUniversal, Fully Scalable Transfection Platform for Production of Complex or Difficult-to-Express Proteins: Superior Performance over Other Transfection Methods & Expression Systems
Biotherapeutic development often requires the production of gram level quantities of recombinant proteins.
Read MoreUsing the Right Cell for the Right Application: Expanding the Role of Transient Transfection
Pharmaceutical, biopharmaceutical, and biotechnology companies must efficiently identify, develop, and quickly bring to market candidates — whether small molecule drugs, therapeutic proteins, or vaccines — with the highest level of efficacy at the lowest cost.
Read MoreMesothelin-Specific Chimeric Antigen Receptor mRNA-Engineered T Cells Induce Anti-Tumor Activity in Solid Malignancies
Off-target toxicity due to the expression of target antigens in normal tissue represents a major obstacle to the use of chimeric antigen receptor (CAR)-engineered T cells for treatment of solid malignancies. To circumvent this issue, we established a clinical platform for engineering T cells with transient CAR expression by using in vitro transcribed mRNA encoding a CAR that includes both the CD3-ζ and 4-1BB co-stimulatory domains.
Read MoreModifying Stem Cells for Clinical Regenerative Applications
Various stem cells types are being evaluated for the potential regeneration of heart, bone, cartilage, nerve and other tissues.
Read More