Rethinking Partnerships in Cell & Gene Therapy—How MaxCyte’s Business Model Drives Real Results
In the fast-moving world of cell and gene therapy (CGT), technology alone isn’t enough. We sat down with Sean Menarguez, senior director of business development at MaxCyte, to discuss how the company’s unique partnership model is helping developers accelerate progress, reduce risk and bring transformative therapies to market faster.
Read MoreCell Therapy eBook SeriesExploring Electroporation 3: iPSCs
Induced pluripotent stem cells (iPSCs), with their unique ability to become virtually any other cell type, are key to unlocking new frontiers in disease modeling, personalized medicine and allogeneic regenerative therapies. iPSCs have tremendous potential to transform human health. MaxCyte electroporation technology, a non-viral cell engineering method, can drive this transformation. It delivers efficiently while maintaining high cell health, saving valuable time and reducing costs.
Read MoreCell Therapy eBook SeriesExploring Electroporation 2: Genetic Diseases
With the growing arsenal of genome editing tools, precise manipulation of the genome is now possible, giving rise to gene therapies that have the potential to cure genetic disease. Explore how MaxCyte electroporation efficiently and reproducibly delivers CRISPR, RNPs and RNAs for targeted gene correction with the flexibility to support today’s research and satisfy the stringent demands of tomorrow’s clinical use.
Read MoreCell Therapy eBook SeriesExploring Electroporation 1: Immunotherapy
Navigate the challenges of immunotherapy development with MaxCyte. As our understanding of cancer biology advances, researchers have responded with increasingly sophisticated approaches to genome engineering. From multiplex targeting of cell surface receptors and immune checkpoint inhibitors to innovative approaches to homology-directed repair, unlock the promise of NK and T cell engineering.
Read MoreMaxCyte® Enabled Mammalian Display Method for the Development of Enhanced Affinity TCR-Based Biologics
MaxCyte electroporation enabled the development of a Flp landing pad CHO cell line, the generation of a TCER CHO library and the production of biologically active soluble TCER candidates by transient gene expression.
Read MoreSeQure DX™ Enabled Pre-Clinical Risk Assessment of PCSK9 Guide RNAs
Find a complete workflow from Guide Profiler to ONE-seq for guide prioritization, selection and off-target nomination for a series of therapeutically relevant guide RNAs targeting the PCSK9 gene.
Read MoreA Novel Cell-Based Assay for Ubiquitin Drug Discovery
Ubiquitin (Ub) is an abundant regulatory polypeptide involved in most cellular processes, added to proteins through a process known as ubiquitylation. The dysregulation or subversion of ubiquitin pathways has been implicated in conditions including viral infection, hereditary neurodegenerative diseases and cancer.
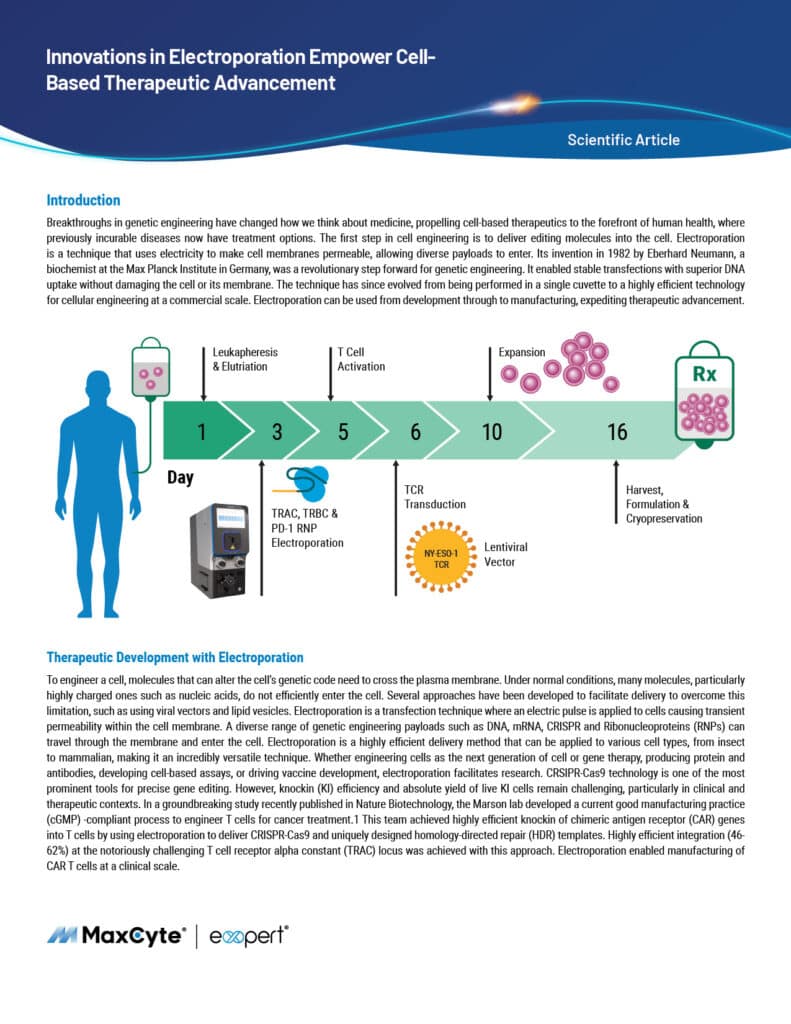
Read MoreInnovations in Electroporation Empower Cell- Based Therapeutic Advancement
Breakthroughs in genetic engineering have changed how we think about medicine, propelling cell-based therapeutics to the forefront of human health, where previously incurable diseases now have treatment options. The first step in cell engineering is to deliver editing molecules into the cell. Electroporation is a technique that uses electricity to make cell membranes permeable, allowing diverse payloads to enter. Electroporation can be used from development through to manufacturing, expediting therapeutic advancement.
Read MoreHigh-Throughput Screening of Ion Channel Variants Using Automated Patch Clamp Recordings in Assay-Ready Cells
In the ground-breaking study summarized here, the authors presented a method for the high-throughput evaluation of the functional consequences of KCNQ2 ion channel variants.1 The technique was used to characterize 39 previously unstudied epilepsy-associated Q2 variants and their responses to treatment with a candidate therapeutic.
Read MoreElectroporation of a Non-Integrative DNA Nanovector for Efficient, Semi-Automated, GMP Manufacturing of CAR T Cell Therapies
Chimeric Antigen Receptors (CARs) have shown extraordinary efficacy in numerous clinical trials as an adoptive cell therapy to treat hematological malignancies. Still, CAR T therapy faces significant challenges, ranging from long lead times and expensive manufacturing to complicated vector engineering.
Read More