Development of a High Yielding Bioprocess for a Pre-fusion RSV Subunit Vaccine
Maxcyte Electroporation helped researchers at the NIH to engineer DG44 cells for a challenging co-expression of two proteins in a respiratory virus vaccine candidate. The resultant clone exhibited high protein stability and expression, producing a 10-fold boost in potentially therapeutic antibodies in just 14 days.
Read MoreDevelopment of anti-human mesothelin-targeted chimeric antigen receptor (CAR) messenger RNA (mRNA)-transfected peripheral blood lymphocytes (CARMA-hMeso) for ovarian cancer therapy
CD19-targeted chimeric antigen receptor (CAR) engineered T/natural killer (NK)-cell therapies can result in durable clinical responses in B-cell malignancies. However, CAR-based immunotherapies have been much less successful in solid cancers, in part due to “on-target off-tumor” toxicity related to expression of target tumor antigens on normal tissue.
Read MoreDevelopment of Enhanced Potency Immunotherapy Products Using Nonviral Approaches
In the next 5–10 years we could see cellular-based pharmaceuticals, or cell therapy, meeting the unmet medical needs of thousands of people. How this therapy will meet these needs depends on the ability of researchers and manufacturers to successfully and cost effectively manufacture and deliver engineered cell-based therapeutic products that are safe and exhibit enhanced potency with resulting durable, meaningful clinical efficacy.
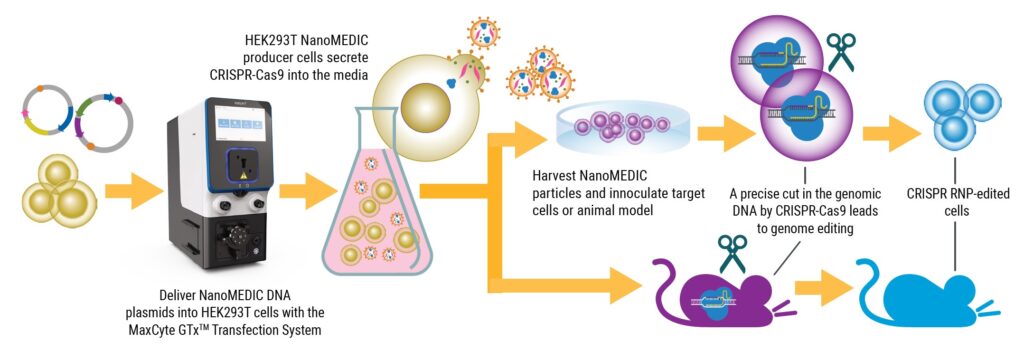
Read MoreLarge-Scale Engineering of Extracellular Nanoparticles for Genome Editing with CRISPR-Cas9 Ribonucleoproteins
Despite advances in human genetics, patients with rare diseases such as Duchenne muscular dystrophy (DMD) still have limited therapeutic options and a short life expectancy.
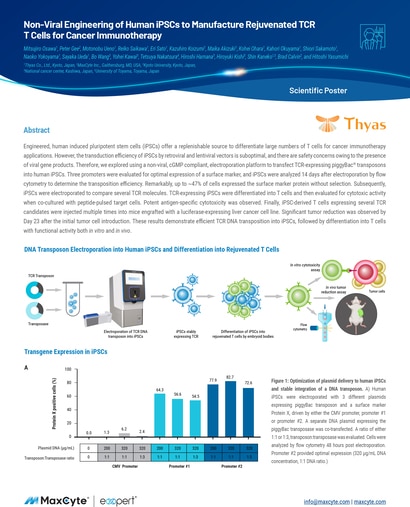
Read MoreNon-Viral Engineering of Human iPSCs to Manufacture Rejuvenated TCR T Cells for Cancer Immunotherapy
Engineered, human induced pluripotent stem cells (iPSCs) offer a replenishable source to differentiate large numbers of T cells for cancer immunotherapy applications.
Read MorePreclinical Optimization of Gene Editing for Adoptive T Cell Therapy with High Fidelity CRISPR/Cas9 Ribonucleoproteins
Adoptive cell therapy with T lymphocytes harnesses the immune system for cancer treatment. T lymphocytes engineered with transgenic T cell receptors (TCR) specific for tumor antigens are in clinical development for various cancers.
Read MoreMaxCyte® Enables Development of New, Rapid, Sensitive SARS-CoV-2 Antibody Test
MaxCyte Flow Electroporation® technology helped researchers develop and implement a new diagnostic test for SARS-CoV-2 infection that is quick, sensitive and easily adaptable as novel pathogenic strains emerge.
Read MoreMaxCyte® Enables a cGMP-Compatible Manufacturing Process for Highly Efficient T Cell Engineering
Excitement over recent breakthroughs in autologous cellular therapies has been tempered by the expense of manufacturing with viral gene delivery and concerns over random integration and the safety of viral vectors.
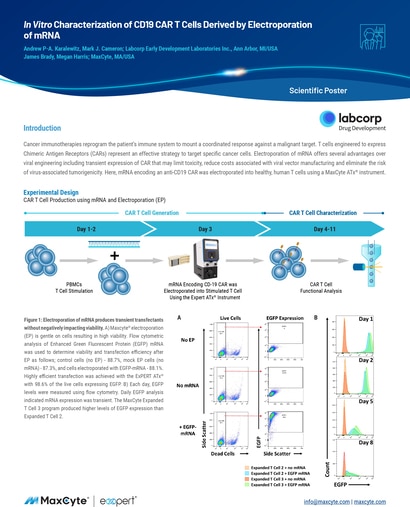
Read MoreIn Vitro Characterization of CD19 CAR T Cells Derived by Electroporation of mRNA
Cancer immunotherapies reprogram the patient’s immune system to mount a coordinated response against a malignant target.
Read MoreMaxCyte® Flow Electroporation® for Gram-Scale Transient Antibody Production in CHO-S Cells
Discover how MaxCyte® Electroporation, a fully scalable technology, enables high-titer antibody production by transient gene expression in CHO-S cells without requiring specific expression constructs, adapted CHO lines, specialized reagents or media additives. Explore how small-scale electroporation was used to identify optimal conditions that could be applied to large-scale transfection without loss of productivity or the need for further optimization.
Read More