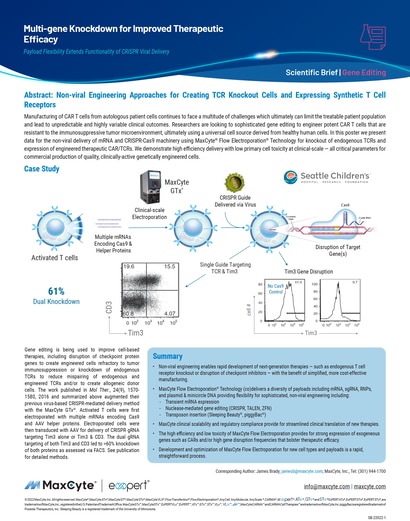
Multi-gene Knockdown for Improved Therapeutic Efficacy
Manufacturing of CAR T cells from autologous patient cells continues to face a multitude of challenges which ultimately can limit the treatable patient population and lead to unpredictable and highly variable clinical outcomes.
Read MoreKeys to Successful Cell-Based Assay Development with Scalable Electroporation
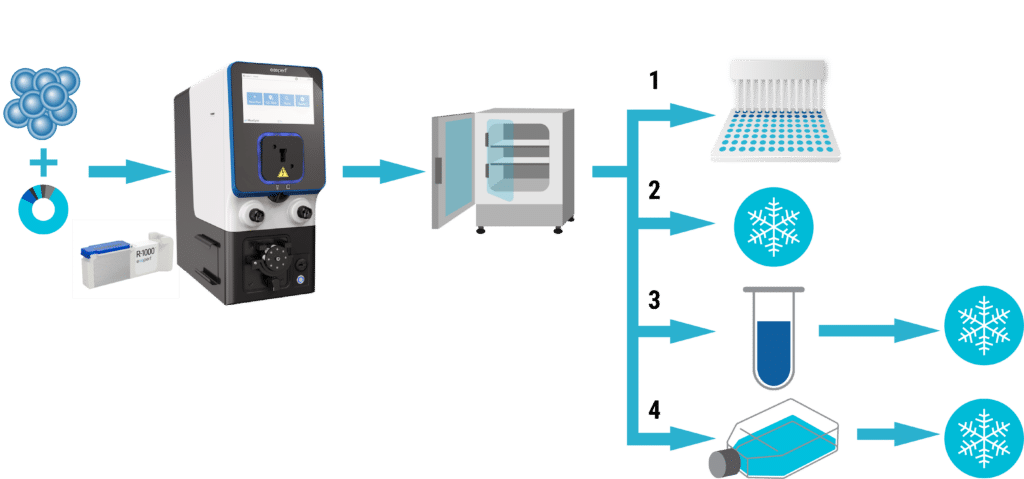
MaxCyte® is a pioneer in electroporation technology for mammalian cell engineering, combining optimized protocols, reagents and consumables to deliver reproducible performance with minimal cell disturbance. MaxCyte’s static and Flow Electroporation® enable high transfection efficiency in virtually any cell type, with seamless scalability from 75,000 to 200 billion cells. MaxCyte’s electroporation technology can be used to produce Assay Ready Cells, addressing the time and cost of assay development with stable cells. The use of Assay Ready Cells facilitates the expression of toxic proteins and enables assay development in physiologically relevant cell types, including primary cells, stem cells and cells of hematopoietic origin.
Read MoreMaxCyte® Flow Electroporation Enables Immunocytokine Development
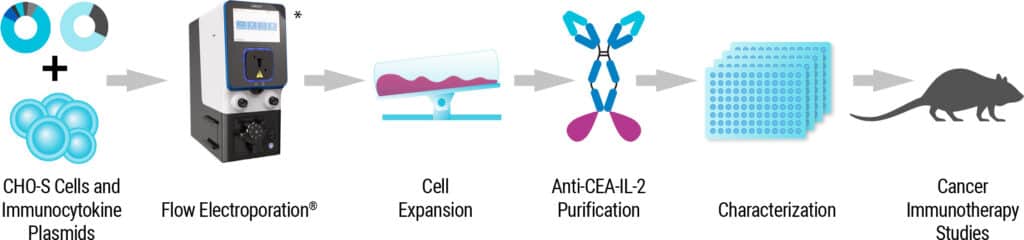
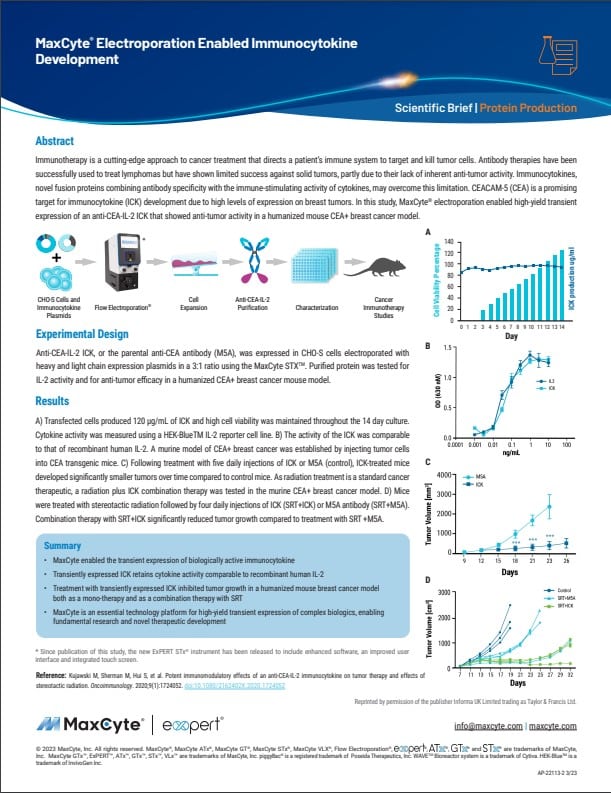
“A novel class of fusion proteins called immunocytokines (ICKs) are an exciting new cancer therapy. They combine antibody specificity with the therapeutic efficacy of cytokines. Learn how MaxCyte® Flow Electroporation® technology streamlined ICK manufacturing and is helping accelerate it into the clinic.”
Read MoreCRISPR Electroporation of T Cells Improves Treatment in Patients with Refractory Cancer
Cancer immunotherapy is a fast-moving field that blends cutting-edge technologies to overcome past obstacles in the clinic.
Read MoreMaxCyte® Enables Rapid, High-Yielding, Stable Cell Line Development
MaxCyte’s highly efficient Flow Electroporation® has extended the potential use of transient protein production in CHO cells as a tool for biotherapeutic development. However, stable monoclonal CHO cell lines remain the system of choice for manufacturing clinical-grade biologics. Stable cell line development continues to be a bottleneck; a time-consuming, costly and labor-intensive process, often requiring specialized equipment. Here, we present a method for generating high-yielding stable CHO clones within six weeks of transfection.
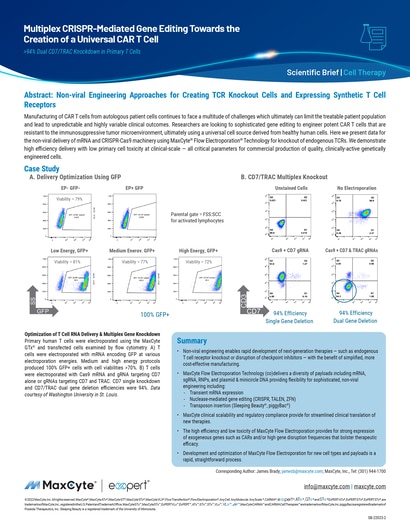
Read MoreMultiplex CRISPR-mediated Gene Editing Towards the Creation of a Universal CAR T Cell
Manufacturing of CAR T cells from autologous patient cells continues to face a multitude of challenges which ultimately can limit the treatable patient population and lead to unpredictable and highly variable clinical outcomes.
Read MoreMaxCyte® Electroporation Enabled Immunocytokine Development
“Immunocytokines (ICKs) are novel fusion proteins combining antibody specificity with the immune-stimulating activity of cytokines. Discover how MaxCyte® electroporation enabled high-yield transient expression of an ICK that showed anti-tumor activity in a humanized mouse CEA+ breast cancer model.”
Read MoreMaxCyte Enables Delivery of Transposons for Highly-Efficient CAR T Cell Engineering
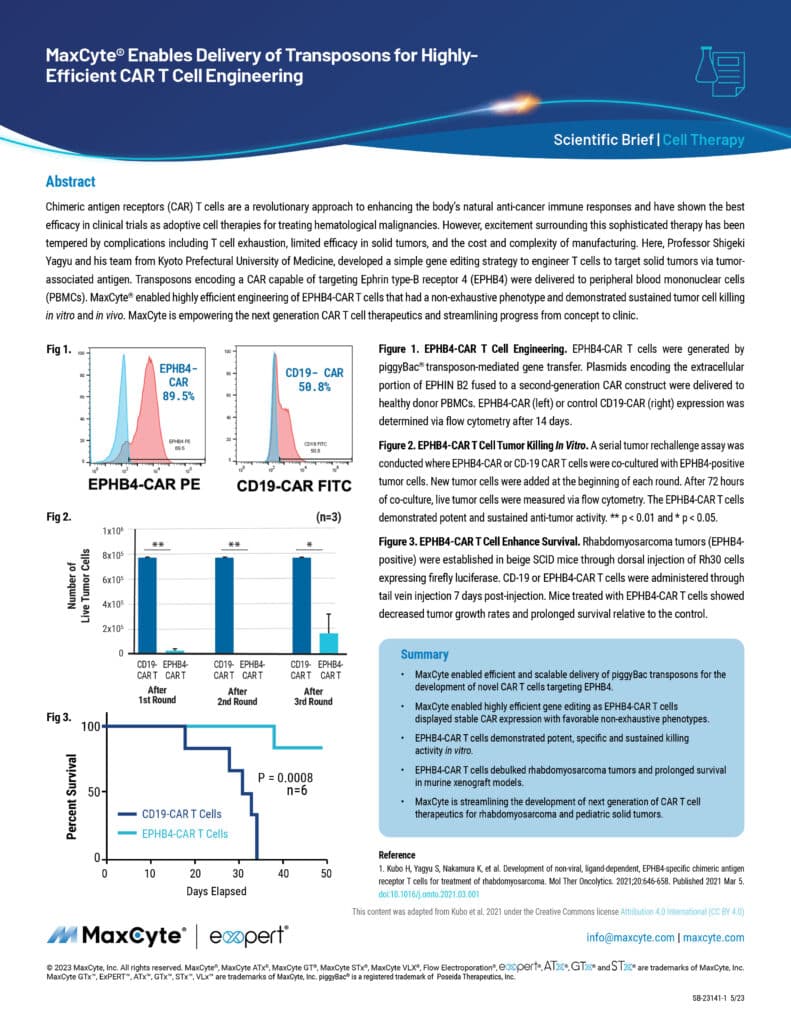
Chimeric antigen receptors (CAR) T cells are a revolutionary approach to enhancing the body’s natural anti-cancer immune responses and have shown the best efficacy in clinical trials as adoptive cell therapies for treating hematological malignancies. However, excitement surrounding this sophisticated therapy has been tempered by complications including T cell exhaustion, limited efficacy in solid tumors, and the cost and complexity of manufacturing.
Read MoreA Fully Optimized CRISPR Workflow for Drug Discovery in T Cells
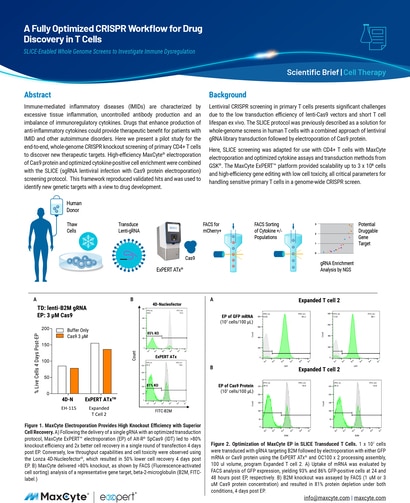
Immune-mediated inflammatory diseases (IMIDs) are characterized by excessive tissue inflammation, uncontrolled antibody production and an imbalance of immunoregulatory cytokines.
Read MoreHematopoietic Stem Cell Electroporation for HDR-Mediated Correction of an X-Linked Blood Disorder
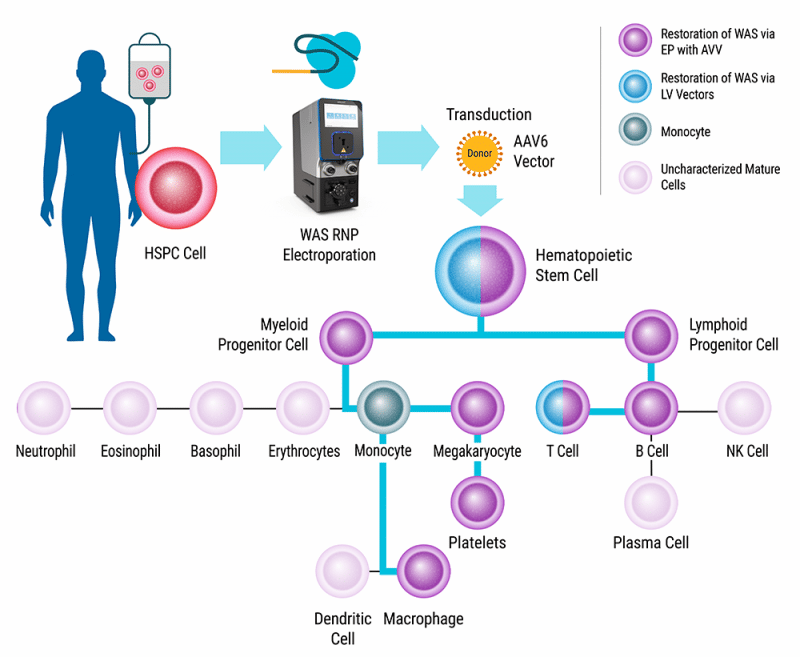
Wiskott-Aldrich Syndrome (WAS) is an X-linked recessive primary immunodeficiency affecting between 1 in 50,000 to 1 in 250,000 live births worldwide. Mutations in the WAS gene cause abnormal immune system function, thrombocytopenia, and an increased incidence of autoimmune malignancies.
Read More